Nuclear fission products11/21/2023  
Where Z is the atomic number and A is the mass number (e.g., Z 2/ A = 36 for uranium-235). The liquid drop model predicts approximately that spontaneous fission can occur in a time short enough to be observed by present methods when Hence, the spontaneous fission of these isotopes is usually negligible, except in using the exact branching ratios when finding the radioactivity of a sample of these elements, or in applications that are very sensitive to even minuscule numbers of fission neutrons (such as nuclear weapon design). The known elements most susceptible to spontaneous fission are the synthetic high-atomic-number actinides and transactinides with atomic number 100 onward.įor naturally occurring thorium-232, uranium-235, and uranium-238, spontaneous fission does occur rarely, but in the vast majority of the radioactive decay of these atoms, alpha decay or beta decay occurs instead. 232Th, 235U, and 238U are primordial nuclides and have left evidence of undergoing spontaneous fission in their minerals. These are nuclei at least as heavy as thorium-232-which has a half-life somewhat longer than the age of the universe. Spontaneous fission occurs over practical observation times only for atomic masses of 232 atomic mass units or more. 
Spontaneous fission was identified in 1940 by Soviet physicists Georgy Flyorov and Konstantin Petrzhak by their observations of uranium in the Moscow Metro Dinamo station, 60 metres (200 ft) underground. Cosmic rays can be reliably shielded by a thick layer of rock or water. Because cosmic rays produce some neutrons, it was difficult to distinguish between induced and spontaneous events. The first nuclear fission process discovered was fission induced by neutrons. 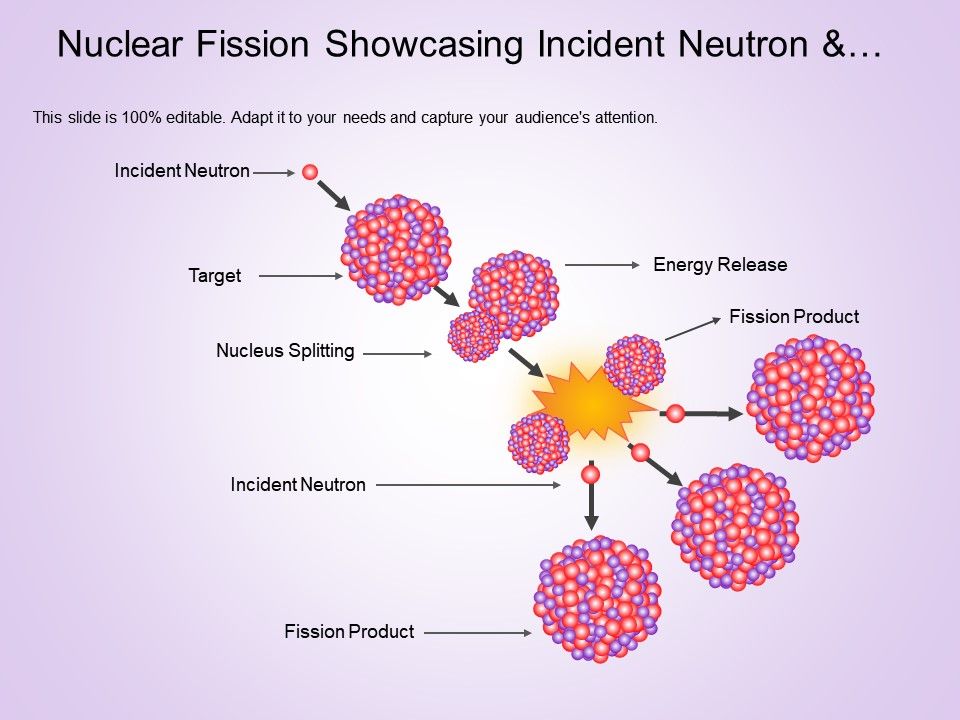
Like cluster decay, alpha decay is not typically categorized as a process of fission. History īy 1908, physicists understood that alpha decay involved ejection of helium nuclei from a decaying atom. The nuclear binding energy of the elements reaches its maximum at an atomic mass number of about 56 (e.g., iron-56) spontaneous breakdown into smaller nuclei and a few isolated nuclear particles becomes possible at greater atomic mass numbers. Spontaneous fission (SF) is a form of radioactive decay that is found only in very heavy chemical elements. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
